Cerium Metal Ce Rare Earth
Product Details:
| Place of Origin: | PRC |
| Brand Name: | HB |
| Certification: | ISO, GB |
| Model Number: | Ce |
Payment & Shipping Terms:
| Minimum Order Quantity: | 2kg |
|---|---|
| Price: | USD6/kg |
| Packaging Details: | Vaccum packed in drums or in wooden case bunddled on pallet |
| Delivery Time: | 5~8 work days |
| Payment Terms: | T/T |
| Supply Ability: | 1t/month |
|
Detail Information |
|||
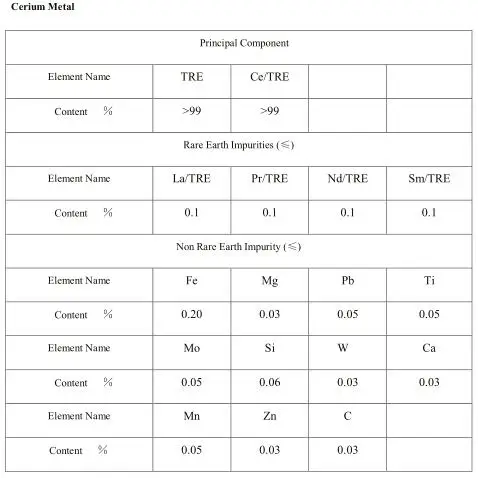
| Product Type: | Rare Earth Metal | Content(percent): | 99% |
|---|---|---|---|
| Name, SymbolOrder Number: | Cerium, CE, 58 | Series: | Lanthanides |
| Group, Period, Block: | La, 6, F | Appearance: | Silvery White |
| CAS Number: | 7440-45-1 | Mass Fraction Of The Earth's Envelope: | 43ppm |
| Atomic Mass: | 140.116 U | Atomic Radius: | 185 Pm |
| Covalent Radius: | 204 Pm | Elektronenkonf.: | [Xe] 4f1 5d1 6s2 |
| 1. Ionization: | 534.4 KJ / Mol | 2. Ionization: | 1050 KJ / Mol |
| 3. Ionization: | 1949 KJ / Mol | 4. Ionization: | 3547 KJ / Mol |
| Physical State: | Fixed | Crystal Structure: | Cubic Area-centered |
| Density: | 6.773 G / Cm3 (25 ° C) | Mohs Hardness: | 2.5 |
| Magnetism: | Paramagnetic (χm = 1.4 · 10-3) | Melting Point: | 1068 K (795 C) |
| Boiling Point: | 3633 K (3360 C) | Molar Volume: | 20.69 * 10 (-6) M (3) / Mol |
| Heat Of Vaporization: | 350 KJ / Mol | Heat Of Fusion: | 5.5 KJ / Mol |
| Speed Of Sound: | 2100 M / S At 293.15 K | Electric Conductivity: | 1.35 * 10 (6) A / (V * M) |
| Thermal Conductivity: | 11 W / (m * K) | ||
| High Light: | Cerium Metal Ce Rare Earth,99 Cerium Metal Ce Rare Earth,Cerium Metal Hydrogen Storage Material |
||
Product Description
Cerium Metal Ce Rare Earth Hydrogen Storage Material
Cerium is the most abundant of the rare earth elements, making up about 0.0046% of the Earth's crust by weight. Commercially cerium and its compounds are being used in catalysts, additives to fuel to reduce emissions and in glass and enamels to change their color.
A traditional use of cerium was in the pyrophoric mischmetal alloy used for flints. Because of the high affinity of cerium to sulfur and oxygen, it is used in various aluminium alloys, and iron alloys. In steels, cerium degasifies and can help reduce sulfides and oxides content, and it is a precipitation hardening agent in stainless steel. Adding cerium to cast irons opposes graphitization and produces a malleable iron. Addition of 3–4% of cerium to magnesium alloys, along with 0.2 to 0.6% zirconium, helps refine the grain and give sound casting of complex shapes. It also adds heat resistance to magnesium castings. Cerium alloys are used in permanent magnets and in tungsten electrodes for gas tungsten arc welding.
Usage:
In metallurgy, cerium is used as an additive for aluminum alloys and high-temperature-resistant iron-based alloys. It supports the separation of sulfur and oxygen in the melting process. The iron-mischmetal alloy Cereisen serves as the starting material for flints for use in lighters and for generating sparks on roller coasters and in movie scenes (accident scenes). Cereisen in the composition 70% cerium and 30% iron, also known as Auermetall, was registered for a patent by Karl Auer von Welsbach in 1903. A modification found worldwide distribution as a flint for lighters.
Small additions of (more or less pure) cerium compounds impart certain properties to other materials:
- Ceria (CeO 2) is used to stabilize the alumina ceramic catalyst support for automotive exhaust catalysts.
- Part of some special lenses, for example UV filters and windshields, and dehumidifiers in glass manufacturing
- For coloring enamel
- Ceria is used as a polishing agent in glass processing
- Cerium-doped fluorescent dyes (phosphors) in picture tubes and white LEDs
- as doping in mantles
- Self-cleaning ovens contain a cerium-containing coating
- Cerium (IV) sulfate as oxidizing agent in Quantitative Analysis (Cerimetry)
- as a contrast agent at nuclear resonance
- as a phosphor in gas discharge tubes
- added to the regeneration of soot particle filters dissolved in the fuel
- as part of non-precious metal-containing bonding alloys in dental technology (ceramics)
- as an oxidant for organic syntheses with CAN (cerium ammonium nitrate), (NH4) ²Ce (NO³) 6

70 Chezhan North Road,Changsha, China 410100
Tel:+86-731-85713359 Fax:+86-731-85716569
Wechat/Whatsapp: +86-185-0731-5452
chris@hbnewmaterial.com chrishuang@vip.qq.com






